FUNDY ISSUES #13
Spring 1999
KEYSTONE COROPHIUM
Master of the Mudflats
"Over the last two decades we have learned
enough about Corophium volutator
to affirm that it is indeed a "keystone species" on the mudflats of the upper
Bay of Fundy. "
Foraging the Fundy flats
When the tide is out, the vast exposed mudflats rimming the upper Bay of Fundy often appear to be a lifeless, monotonous wasteland of little interest to anyone or anything. Yet, for a short five or six weeks each year the flats come alive with huge flocks of migrating shorebirds, obsessively probing the sticky wet mud. Chief among them are Semipalmated Sandpipers, making a brief but critical feeding stop on a 5,000 kilometre journey from subarctic breeding grounds to tropical wintering grounds. The 2-3 million birds that visit Fundy represent 75 to 95% of the world’s population. They come seeking a tiny, elusive, burrowing amphipod, Corophium volutator, a nutritional mainstay for the last, non-stop leg of their journey south. The story of the sandpipers and their affinity for these seemingly inhospitable mudflats is told at greater length in Fundy Issues #3, "Sandpipers and sediments: shorebirds in the Bay of Fundy". Occasionally, small flocks of larger two-legged waders can also be seen foraging on the flats, probing the mud as obsessively as any sandpiper. They too have a voracious appetite for Corophium, but an appetite stimulated by curiosity rather than hunger. These are flocks of scientists and their broods of fledgling students, many associated with the Acadia Centre for Estuarine Research. ACER has spearheaded studies on the ecology of Fundy’s mudflats and salt marshes for more than two decades. Corophium volutator is also found from Scandinavia to the Mediterranean, and there has been speculation that it may have been accidentally introduced in the Bay of Fundy and Gulf of Maine following European settlement. A preliminary comparison of their genetic "fingerprints" suggests, however, that the populations have been separated far longer. This is a synthesis of what the flocks of dedicated researchers on both shores of the Atlantic have learned about the subtle, delicate and finely-balanced interplay that exists among species and with the glutinous habitat that they share. Their studies show that Corophium is not only incredibly abundant and productive on many mudflats, but may also be a "keystone species". Just as a strategically placed keystone supports the structural integrity of a stone arch, so a "keystone species" shapes and sustains the ecological integrity of its surroundings. Remove the keystone and an arch becomes a heap of stone; remove the keystone species and an ecosystem changes its structure and productivity.
Amphipod anatomy Corophium is called a "mud shrimp" locally; but it isn’t a shrimp, rather a member of a diverse suborder of crustaceans named "amphipods". The best known are "scuds" or "sideswimmers" that scuttle around erratically when damp seaweed on the lower shore is lifted, and "beach fleas", that leap about in great confusion when the windrows of decaying seaweeds festooning the upper beach are disturbed. All amphipods have a stiff chitinous armour or "exoskeleton" similar to that of lobsters. As they grow they must periodically cast off or "moult" the exoskeleton and form a roomier one. Unlike lobsters, amphipods do not have an elongated, one-piece casing or "carapace" to protect the forward half of their body, the "thorax". Instead their entire body is divided into a series of short, tubular segments. Sturdy, flexible joints between segments allow the animal to straighten or bend into a ball. Typically each segment has a pair of jointed limbs. Along the body, small groups of these limbs are modified in size and shape to perform many different functions. The head bears two pairs of long, flexible antennae, originally designed for sensing the environment. However, Corophium also uses them for other things, as we shall soon see. Behind the antennae, on the underside of the body, is a grouping of small limbs chiefly used in the capture, handling and ingestion of food. These vary slightly in size and shape according to their specific role in feeding, and are called "maxillae", "maxillipeds" and "gnathopods". Within the mouth, paired, hard protrusions or "mandibles" forcefully grind the food. Small, thin-walled flaps or "gills" attached at the base of some of the feeding limbs absorb oxygen from water flowing over them. Next along the "thorax" are several long, flexible walking legs or "periopods" used for crawling about on the seafloor. Tucked beneath the rear half of the body or "abdomen" are of a series of small, feather-like limbs ("pleopods"). These beat in unison like miniature paddles and provide the thrust for occasional swimming, while at a slower pace they create a current of oxygenated water over the gills. The broad flat tail segment ("telson") and pairs of leaf-like limbs ("uropods") together form a broad tail fan similar to that of a lobster. When the abdomen is suddenly flexed forward, this tail paddle drives the animal swiftly backwards, a handy defensive manoeuvre when predators strike. Corophium is small compared to most amphipods, ranging in length from about 1 mm when first hatched, up to 12 mm (excluding the long antennae), the largest reported in the Bay of Fundy. Juveniles mature and sexes become readily distinguishable at about 5 mm in length; the most striking difference being the longer, sturdier-looking second antennae in males.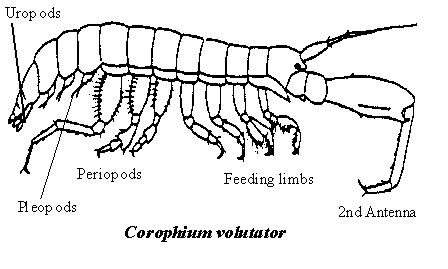 |
Corophium keeps a low profile; so low in fact that until a couple of decades ago the mudflats of Fundy were considered lifeless wastelands of little ecological interest. But as sandpipers have known for innumerable generations, and scientists have recently learned, if you probe beneath the surface, the mud is home to unbelievably large numbers of these tiny amphipods. To most of us a mudflat is a mudflat, and a rather boring, barren, lifeless, featureless expanse of real estate at that. But Corophium is more discriminating in its perception, and over the flat’s vast, unmarked expanse there are clearly desirable residential neighbourhoods and others to avoid. The salt content of the water washing over the flat is a minor consideration. Corophium can live in water saltier than seawater (~34 parts per thousand or "ppt." of salt) as well as in water only slightly saltier (~5 ppt. of salt) than freshwater, and readily tolerates everything in between. However, given the choice they tend to prefer, and thrive best at, a salt content around the middle of this range (~20 ppt. of salt).
Laboratory studies by Dr. Meadows in Wales shows
that Corophium is "apparently quite deliberate" in choosing where it burrows.
They aren’t simply attracted from afar to a particular area, but rather swim about at
random, alighting occasionally to check out the surface. The colour of the substrate
isn’t a concern, for they can pi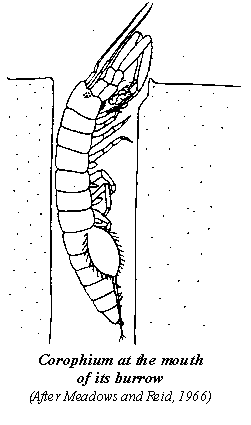 ck out suitable sites even in the dark. Upon
alighting, they crawl about and carefully manipulate particles of sediment, possibly to
assess their texture and chemical characteristics. They respond to "some property of
the surface layer of substrate when deciding whether or not to burrow". The average
house-hunting Corophium is chiefly concerned about the size and nature of the
sediment particles present in different parts of the mudflat. Ideally they should be of a
particular size range, with just the right mix of silt, clay and sand; neither too finely
silty, nor too coarsely sandy. It doesn’t mind a little blackish mud with low oxygen
just beneath the surface. In fact, in the laboratory, if offered a choice it opts for
sediments with reduced oxygen content. The size of particles is particularly important for
the mud to have the right consistency for digging burrows. If it is too fine and watery,
burrows can’t be built or soon collapse. If it is too coarse, less organic film is
present on the surfaces of the large particles. Sediment particles must be small and well
coated with such organic matter if they are to be acceptable as food. These natural slimy
"biofilms" typically consist of bacteria, microscopic algae, fungi, protozoans
and their secretions, as well as other organic compounds that stick to the particle
surface. As we shall see, such biofilms are important as food; so Corophium is not
just looking for a good stable home, but one with a well-stocked larder as well. In
addition, the depth of the sediment layer must be at least ~0.5 cm, with larger animals
requiring an even deeper layer in which to burrow.
ck out suitable sites even in the dark. Upon
alighting, they crawl about and carefully manipulate particles of sediment, possibly to
assess their texture and chemical characteristics. They respond to "some property of
the surface layer of substrate when deciding whether or not to burrow". The average
house-hunting Corophium is chiefly concerned about the size and nature of the
sediment particles present in different parts of the mudflat. Ideally they should be of a
particular size range, with just the right mix of silt, clay and sand; neither too finely
silty, nor too coarsely sandy. It doesn’t mind a little blackish mud with low oxygen
just beneath the surface. In fact, in the laboratory, if offered a choice it opts for
sediments with reduced oxygen content. The size of particles is particularly important for
the mud to have the right consistency for digging burrows. If it is too fine and watery,
burrows can’t be built or soon collapse. If it is too coarse, less organic film is
present on the surfaces of the large particles. Sediment particles must be small and well
coated with such organic matter if they are to be acceptable as food. These natural slimy
"biofilms" typically consist of bacteria, microscopic algae, fungi, protozoans
and their secretions, as well as other organic compounds that stick to the particle
surface. As we shall see, such biofilms are important as food; so Corophium is not
just looking for a good stable home, but one with a well-stocked larder as well. In
addition, the depth of the sediment layer must be at least ~0.5 cm, with larger animals
requiring an even deeper layer in which to burrow.
The composition of the mud in different parts of a flat can vary greatly. Current or wave action may wash out fine particles in some areas, while elsewhere small depressions or channels may trap fine sediments and organic material. Mudflats thus tend to be a patchwork, and it is not surprising that the distribution and abundance of Corophium is also patchy. This makes it difficult to estimate numbers on a particular mudflat. The patchiness is further enhanced by the fact that Corophium likes to be close to its neighbours; the presence of other burrows is a bonus in site selection. In the laboratory, animals overwhelmingly preferred mud containing previously burrowed animals to mud without burrows. Interestingly, groups of animals burrowed readily, while isolated ones did not. In selecting sites, the animals also check the depth by probing the substrate with their elongated second antennae. If conditions are suitable they burrow and if not, they simply move off to investigate other sites.
Each tiny, U-shaped burrow has two openings to the sediment surface. Its size varies with that of the inhabitant, but is usually a few millimetres in diameter and extends down into the mud some 7-cm or more, for a total length up to 15-cm The volume of the burrow of an average adult has been estimated to be about 0.5 ml. The animal carefully burrows in the soft mud, using its large antennae and periopods to loosen the sediment and its beating pleopods to waft the excavated material away. The walls of the burrows are compacted sediment particles bound together with an adhesive secretion from special glands. The animals do not remain permanently in one burrow, but move frequently, sometimes every day or two. Animals in laboratory tanks occasionally emerge from the mud to explore the surface, and soon either move into a vacant burrow or construct a new one. There appears to be no lack of available real estate, as sometimes more than half of the burrows in an area can be vacant. However, as population density increases, just as in any urban centre, occupancy rates rise and vacancies become scarce. If a prospective tenant blunders into an occupied burrow a scuffle usually ensues, and in the words of the researcher, "the original occupant, irrespective of its size, usually manages to throw the intruder out". It seems that possession is nine-tenths of the law for Corophium as well. However, as we shall see, if the occupant is of the opposite sex then the outcome may be quite different.
Managing mudflats
Given their tiny size, Corophium have a surprising influence on the ability of mudflats to withstand the ceaseless eroding forces of currents and waves. Diatoms, bacteria and other micro-organisms in the surface layers of mud secrete sticky, organic substances called "muccopolysaccharides". This glue-like material makes the sediment particles stick together and reduces the likelihood of their being swept away by moving water. As we shall see, Corophium consumes large numbers of these tiny organisms, resulting in a drop in the amount of sticky stuff produced. As a result the sediment particles don’t stick as much and are more easily washed away. This has been demonstrated by spraying an area of mudflat with insecticide to kill off the Corophium. The diatoms and bacteria flourished, and the sediment’s ability to resist erosion increased significantly. Flocks of hungry shorebirds eating large numbers of Corophium can have a similar stabilising effect on a mudflat. Graham Daborn and his colleagues call this an "ecological cascade", with shorebirds influencing Corophium abundance, which in turn influences diatom abundance, which in its turn influences the stability of the mudflat sediments. This ecological linkage is described in more detail in Fundy Issues Number 3, "Sandpipers and sediments: shorebirds in the Bay of Fundy". However, Corophium may also have a positive effect on sediment stability. When it builds its burrows it not only compacts the intervening sediment, but also cements together the particles lining the walls, creating a forest of erosion-resistant chimneys in the mud. The manner in which these opposing effects of Corophium on mud stability interact together in the real mudflat world is undoubtedly complex and not fully understood.European studies suggest that Corophium may also unwittingly defend its mudflat habitat from invasions by salt marshes. Mudflats are constantly threatened by the encroachment of nearby salt marshes, which are essentially vegetated mudflats. Pioneer species of marsh plants, such as the glassworts (Salicornia spp.), continually strive to colonise outward from the fringes of the marsh and claim footholds on the mudflats. They stabilise the sediments with their root systems and trap even more sediment, creating conditions that allow a full range of salt marsh grasses and other plants to follow. Corophium actively resists these invasion attempts. Firstly, by consuming diatoms it keeps the mud surface too unstable for colonising plants to establish themselves. Secondly, and more directly, by burrowing and feeding it either buries the seeds of the invaders preventing them from germinating, or uproots any that do. Furthermore, this "mudflat defense" strategy may be especially focussed on the most vulnerable areas. The net shoreward transport of swimming Corophium on rising tides, observed in some European estuaries, tends to concentrate them at the periphery and in the drainage channels of salt marshes. There is thus a constant augmentation of the Corophium population in the very areas where the threat of salt marsh invasion is greatest. Whether this occurs to any significant extent in the upper Bay of Fundy is not known.
Amorous excursionsAlthough some aspects of the reproduction of Corophium are still a mystery, we have learned a great deal. There are usually as many as four to ten times more females than males: what is termed a highly skewed sex ratio. Typically, it is the outnumbered males that initiate reproduction. On ebbing tides, in the short interval after the water has receded, when the sediment surface is still soft and wet, mature males emerge from their burrows and set off in search of a mate. Thanks to painstaking observations by researchers, such as Sherman Boates and colleagues affiliated with Acadia University, we have a clearer picture of these preliminary courtship rites. Far out on a mudflat, for an hour or two as the tide ebbed, these researchers hunched over the mud and diligently followed individual males crawling about in search of females. The amphipods tentatively investigated any burrows they encountered. At the mouth of each one examined, but not entered, the researchers dutifully planted a tiny, red flag. Eventually, after checking several burrows the male would find one to his liking and disappear inside. This burrow was marked with a green flag. As many as ten animals were followed in this manner on each tide. Before the tide returned, the researchers carefully dug up the patch of mud containing all the flagged burrows. They then teased apart the mud to collect the occupants of the burrows, recording the number in each, their sex and size, as well as the reproductive condition of females. In the course of 31 such trips to the mudflat they successfully tracked a total of 236 males and learned several interesting things. Most males visited from 2 to 6 burrows before finally entering one, although a few hard-to-please ones visited as many as 17 or 18 before committing themselves. Occasionally a roving male happened upon the home of another male, or one containing an already paired male and female. Such encounters inevitably led to conflict between the males in the course of which "the tube dwelling male and intruder interlocked their second antennae and the tube dwelling male pushed the intruder away from the burrow". However, many of the burrows visited did contain potential mates. To the researchers’ keen eyes, the "males definitely appeared to assess the females" stationed at the mouths of their burrows. They delicately ran their long antennae back and forth along the underside of the female, sometimes for three minutes or more. This caress may have helped determine the reproductive condition of the female, as males were much more likely to settle down with females on the verge of laying eggs. Surprisingly, given that larger females usually lay more eggs, the males didn’t seem to regard size as important in mate selection, as long as they were ready to lay. Once a male made his choice he moved into the burrow with his new mate. The females didn’t appear to be so discriminating, for there was "no clear rejection of males by females". However, on three separate occasions researchers saw "females grab a male and attempt to drag it into the burrow". They laconically concluded that these were "attempts by females to secure matings from males". They note that this is not surprising given that "one sex is a limiting resource".
The manner in which copulation occurs in Corophium has never been observed, because it takes place in the privacy of the burrow. However, it is undoubtedly similar to that of other less secretive amphipods. An amphipod’s strategy for bearing young is somewhat akin to that of marsupials such as the kangaroo, in that the eggs are deposited into a pouch-like brood sac or "marsupium". Here they are fertilised and develop until the young are old enough to fend for themselves. The marsupium is slung beneath the thorax and is formed by several leaf-like blades or "oostegites" that project inward from each side and overlap in the middle to form the basket-like structure that forms the floor of the marsupium. Typically, copulation occurs soon after a female has moulted. The male twists his abdomen around so that the tail fan, which is near where the sperm ducts open, touches the marsupium. Sperm is released into the water and is immediately swept into the marsupium on the currents produced by the pleopods. The pair separates and almost immediately eggs are released into the marsupium and are fertilised. Females can lay between 10 and 172 eggs, depending on her size, but the average is about 38. It is not clear why Fundy animals appear to lay more eggs than their European cousins. Females carrying eggs or developing embryos in the brood pouch are referred to as "gravid". Most females don’t become gravid until they are larger than about 6 mm in length, although ones as small as 5.4 mm have been found. The round eggs are initially creamy white and surrounded by a thin, flexible "egg membrane". Over a period of about two weeks the eggs divide repeatedly to form embryos in which limbs and eyes soon become visible. All the while, the steadily beating pleopods flushes well-oxygenated water around the developing young. Amphipods undergo "direct development", meaning that the egg hatches out a miniature version of the adult rather than an unfamiliar looking larva. During development, as many as a quarter of the eggs or embryos may disappear from the brood pouch, as a result of some simply dying and others falling out.
Once the embryos are fully developed they are ready to break free of the protective egg membrane. They do this with the aid of "hatching spines" which are two to four sharp projections located on the top edge of the tail segment (telson). Curiously, these are not present on the first embryonic exoskeleton, but are on the new exoskeleton developing beneath it. In effect, the tiny animal has to moult and hatch almost simultaneously so that the newly exposed hatching spines can make an initial tear in the egg membrane as the animal squirms vigorously. A final series of violent contractions are usually sufficient to free the animal completely from the torn egg. Newly hatched animals are a 1.0 mm long replica of the adult. Free-living animals of this size have been collected, suggesting that some leave the marsupium almost immediately. Most, however, remain inside for several days and may even moult again before finally launching out into the world.
The breeding season of Corophium in the upper Bay extends from early May until early August. In addition to this seasonal pattern, reproductive activities are also closely linked to the tidal cycles. As we have already seen, mate searching by males is largely confined to the hour or so after the mud is exposed by the receding tide. But in addition to the twice-daily (semidiurnal) ebb and flow of the tide, there is another longer cycle linked to the waxing and waning of the moon. Each high tide reaches successively higher on the shore until it reaches a maximum height (spring tide, but nothing to do with the season). Then each subsequent high tide decreases gradually to a minimum height (neap tide). This cycle repeats endlessly, with the high spring tides recurring roughly every two weeks. The number of crawling males rises sharply at about the time of each spring tide in the summer, and not surprisingly, so does the number of gravid females. This careful timing ensures that when the fully-developed young are released some two weeks later, the next spring tide will have arrived, providing the juveniles with a much broader expanse of intertidal mudflat than normal to explore and on which to settle down.
Diatoms and dietsCorophium are not finicky eaters, but the food must be of a suitable size. They can feed on particles lying on the seafloor (deposit feeding) as well as on ones suspended in the water (filter feeding). To deposit feed, they partly emerge from the burrow and use the large second antennae as rakes to pull particles towards the mouth of the burrow. Currents produced by the beating pleopods draw in this light, loose material. Some of the feeding limbs, the gnathopods, have long, fine bristles or "setae" which overlap to form a filtering basket through which the water passes. Particles larger about 0.004-mm are trapped on the mesh. Other feeding limbs deftly comb the particles off the mesh, sorting out the desirable ones and moving them forwards to the mouth. Here they are crushed between the grinding mandibles and swallowed. Essentially the same procedure is used to strain out any food particles suspended in the water flowing through the burrow. The pleopods may pump between 25 and 100 ml of water per hour through the burrow, bearing both food and oxygen. After a period of feeding at one opening, the animal withdraws into its burrow and may either rest or dextrously turn around in the narrow confines and emerge headfirst to continue feeding at the other entrance. They deposit feed at any stage of the tide and no daily cycle in their dining is evident.
 |
Corophium may pick at their food, but they certainly don’t dawdle over it. An actively feeding animal can fill its stomach in about half an hour and digestion takes less than an hour. Examination of their stomach’s content offers little insight into the diet of Corophium. Typically less than 1% of the material is identifiable under the microscope, a tribute to the grinding efficacy of the tooth-like mandibles. Bottom deposits and seawater contain a variety of potential food particles of a suitable size (0.004 to 0.060mm). In summer, the most important are cells of single-celled plants or "diatoms" that flourish on the damp surface of the mud ("benthic"). A single animal may consume as many as 4000 diatoms a day. In a year, the Corophium population could consume more than a quarter of the total benthic diatom production of a given area. Decaying fragments of plant material or "detritus", wash off nearby salt marshes onto the mudflats, particularly in the spring and fall and supplement their diets when diatoms are scarce. But a limited ability to digest the plant detritus suggests that they may be feeding mostly on the bacteria growing on it. Bacteria seem to be an important food source so long as they are attached to particles larger than about 0.004 mm; because by themselves they readily pass through Corophium’s filtering mesh. They inevitably ingest a large amount of fine sediment when deposit-feeding and the organic–rich biofilms coating the particles are probably digested during passage through the gut. They can also gnaw off the biofilms from larger particles using their mandibles in a process called "epipsammic browsing". Up to three-quarters of the diatom material, but only about a quarter of the bacterial material, is digested and absorbed. Lipid digesting (lipases) and protein digesting (proteases) enzymes are present in the stomach. Much lower amounts of cellulose digesting enzymes (cellulases) confirm that they can’t directly digest salt marsh detritus very well. The proportions of these digestive enzymes present in the stomach changes from season to season in accordance with the different foods available. Thus in spring Corophium may rely on the more abundant salt-marsh detritus and accompanying bacteria, while in summer they consume chiefly their preferred diatom diet.
Corophium eliminate light, fragile "faecal pellets" that are up to 0.5 mm in diameter and 10 mm long. They are released in the burrow, but are soon flushed out and accumulate near the entrance. An actively feeding animal can expel a pellet every 2 to 4 minutes and in a day 500 to 1000 pellets may be produced. The delicate pellets are quickly dispersed by currents and waves.
Population parametersWhen habitat conditions are right and food is plentiful, Corophium populations can attain incredibly high densities. On a productive mudflat there can be 10,000 to 20,000 animals per square metre (about the size of a beach towel). In some areas of Fundy, densities greater of 60,000 per square metre have been observed. But their numbers are not this high throughout the year. The intricacies of the life cycle and predation by birds and fish result in sharp seasonal peaks and troughs in abundance. However, not all populations follow exactly the same patterns. In the upper Bay of Fundy, two distinct generations are produced each year. In contrast, in the outer Bay, around and below Saint John, N.B., there is only one annual generation. This is thought to stem from the colder average temperature (~6�C) that prevails there. A similar phenomenon occurs in European waters, with single generations in colder northern areas and two in warmer southern climes.
Corophium begin the year as juveniles in their burrows where they have overwintered. The cold itself had not been a problem, although they may have snuggled a bit deeper into the mud when air temperatures dropped. Even if they had been frozen solid into the mud they could have revived upon thawing. However, as indicated below, in exceptionally cold winters, heavy ice may destroy burrows and thus decimate the populations. However, enough animals survive even the harshest winters to ensure that the population prevails. These survivors begin reproduction in early May when gravid females appear. The first young are released in late May. By mid-June, there is a great surge in the number of these small juveniles. A single female may produce more than one brood, further fuelling the population explosion. These spring progeny grow rapidly with the summer warmth and an abundant supply of diatom food, and by early July they begin to mature and become gravid. By the end of July they are releasing their own young in large numbers. It is this late summer, second generation that is destined to struggle through the long winter and begin the cycle anew the following year. They grow slowly through the autumn and cease growth by December, remaining as rather torpid, small juveniles until the promise of spring sets them growing once again in April.
Perils aplentyCorophium faces many perils on its deceptively benign-looking mudflat. An increasing number of these threats bear the imprint of human handiwork, but most are quite natural. Corophium is high on the menu for a wide range of hungry animals. As the rising tide washes over the mudflats, schools of small fish such as tomcod, flounder, smelt and mummichogs, which have waited expectantly just beyond the surf, scurry in over the newly submerged bottom in search of a meal. That this muddy larder is well-stocked is evident from the fact that on the other side of the tide line equally numerous flocks of hungry shorebirds, such as sandpipers, plovers and other waders impatiently await their turn at the same table once the water recedes. Corophium is the main course at both these intertidal banquets. Not much is known about the predation by fish and the effects on the Corophium population, because it occurs hidden beneath the waves. A great deal more is known about the effects of predation by birds, thanks to the decades of intensive studies by Peter Hicklin of the Canadian Wildlife Service in Sackville New Brunswick and Sherman Boates, of the Nova Scotia Department of Natural Resources. Both launched their initial research on Corophium while students at Acadia University.
The annual onslaught by the hungry shorebirds is intense (2-3 million birds, each eating from 10 to 20 thousand Corophium during a single tidal cycle) but relatively brief (about 6 weeks, in late July and early August; although individuals remain only for the 10-15 days needed to double their weight). Researchers think that this highly focussed, short-term attack alters not only the size distribution of the prey but also their population dynamics and possibly even their genetic characteristics. The mudflats at Starr’s Point and Blomidon in the Minas Basin, near Acadia University, are providing an ideal natural laboratory for studying the effects of such heavy predation. The sandpipers feed intensively on Corophium at Starr’s Point. However, only 10 km away at Blomidon, very similar mudflats are largely unvisited, because the high tides wash right up to the steep cliff face, leaving no beach for the birds to roost on while they wait for the tide to turn. The exact nature and ecological implications of the interesting differences in the life history of the Corophium populations at the two sites are only now being explored.
Some of the differences are undoubtedly linked to the fact that the sandpipers are rather choosy as to which Corophium they eat. They clearly select the larger ones, particularly the larger males, which thus bear the brunt of predation. As we have seen, these are the very animals that come out of their burrows as the tide recedes, to scout the mudflat for mates. The sandpipers have learned that by following the receding tide down the flat they will find an abundance of large, amorous amphipods right on the mud surface – easy pickings! But it seems that the amphipods somehow recognise the danger that they are in, for within days of the arrival of the sandpipers, the number crawling on the surface decreases by more than 95%. There are still lots of large males burrowed in the mud, but somehow they sense that sandpipers are on the prowl and simply stay indoors. The sandpipers then have to resort to probing in the mud, where they still seek out the larger adults. Primarily eating the adults is a wise strategy from the bird’s point of view, because it is the uneaten juveniles that will live through the winter and produce the spring brood that will become their sustenance on their trip south the following year. The adults on the other hand are nearing the end of their life, having done their reproductive duty. By consuming them in large numbers the birds may be freeing up valuable habitat space and reducing competition for burrows and food, allowing the juveniles to grow more rapidly once the sandpipers have departed.
 |
But marauding birds aren’t the only threat that Corophium has to worry about. Their habit of dwelling in the surface layers of intertidal, estuarine sediments makes them vulnerable to exposure to a variety of noxious or toxic materials that are washing into the marine environment. Many studies have demonstrated the animal’s sensitivity to some of these substances, such as organic wastes, pesticides, crude oil, heavy metals and PCB’s (polychlorinated biphenyls). These often bind to sediment particles and tend to accumulate in mudflats where they can be ingested or absorbed by the species living there. Corophium is sufficiently sensitive to some of these compounds to warrant being designated by Environment Canada as a standard test organism for assessing how toxic contaminated sediments are to marine life. Many of these compounds are ubiquitous in the marine environment, so it is not surprising that they have been detected in the habitat and tissues of Corophium in many places. At present the concentrations of most such compounds in the mudflats of the upper Bay are low and pose little threat to Corophium. The same cannot be said of mudflats located in estuaries that are dominated by large urban or industrial centres, such as near the city of Saint John, New Brunswick, or adjacent to large aquaculture operations, such as those in and around Passamaquoddy Bay.
Not only may Corophium itself be threatened by the build-up up of contaminants in the sediments, but its burrowing activities may increase the risk of exposure for many other organisms as well. By intensely reworking the surface layer of contaminated mud (a phenomenon known as "bioturbation") Corophium increases not only the amount of suspended matter in the overlying water but also the concentration of the contaminants that are bound to it. This makes the contaminated particles more accessible to animals that feed by filtering the water, such as mussels. The more amphipods there are in an area, the greater the transfer of contaminants to filter feeders. Thus, Corophium can play a significant role in transferring toxic substances buried in bottom sediments into the aquatic food chain.
Perhaps the most worrisome of the threats presently facing Corophium populations are changes in sediment dynamics and mudflat composition. It is suspected that some of the changes may be long-delayed effects of the blockage of most large rivers flowing into the Bay of Fundy by causeways over three decades ago. Additional changes in sediments may result from the already implemented and proposed experimental openings of tidal gates in some causeways. The nature of such sediment changes and the threats that these may pose to the ecosystem are discussed at greater length in Fundy Issues Number 3, "Sandpipers and sediments: shorebirds in the Bay of Fundy" and in Number 9, Dykes, dams and dynamos: the impacts of coastal structures".
A more immediate and localised threat to the habitat of Corophium comes from the widespread and intensive commercial harvest of bloodworms, Glyera dibrachiata, on the mudflats of the upper Bay. Large numbers of the worms are being exported for use as live bait in recreational fisheries, largely in the northeastern U.S. This "fishery" surged northward into the Bay after 1985, following the overexploitation and decimation of baitworm populations in New England waters. Harvesting involves manually turning over the upper 10-20 cm of mud with a fork-like rake. This not only destroys existing burrows, but also loosens the sediment, making it difficult to construct new ones. Large swaths of mudflat can be turned over during a single low tide by a small group of diggers. Such "ploughing" puts large amounts of sediment into suspension and destabilises extensive areas of intertidal mudflat. Corophium densities decline by as much as 40% on such areas. Juveniles are even more susceptible, for their numbers can be cut in half. This also has serious repercussions for the migrating shorebird populations, whose feeding ability can be reduced by two-thirds in these disturbed areas. These adverse effects on Corophium and shorebirds can persist for a year or more after harvesting ceases.
Natural processes can also physically disturb the mudflat habitat. In years when ice conditions are particularly harsh, large numbers of Corophium may be killed, not from the freezing, but from having their burrows destroyed by the ice. The surface of the mud can be deeply scoured and gouged by large, heavy chunks of ice moved around by tides, waves and currents. Sometimes this ice freezes into the mud surface at low tide and then floats off with layers of the mud attached as the tide rises. Major storms, usually prevalent in the autumn, can also kill large numbers of animals as a result of large waves washing away sediments and destroying burrows. Usually, after either harsh icing or storms, the populations recover by the following summer, with animals quickly recolonising from nearby undisturbed areas.
In some European estuaries, excessive amounts of dissolved nutrients adversely affected Corophium populations. Nutrients arising from domestic wastes or agricultural runoff stimulated an overproduction of seaweeds and other algae, a phenomenon termed "eutrophication". As we have seen, Corophium readily handles the tiny benthic algae or diatoms. But the larger algae such as Enteromorpha, or the sea lettuce Ulva, spreading across a mudflat can quickly wipe out a Corophium population. The constant sweeping of the seaweed fronds across the sediment surface disrupts the feeding of the animals and often entangles them. The high tidal flushing makes it unlikely that eutrophication will ever be a problem in most areas of the upper Bay of Fundy, although it could be occurring in estuaries near the mouth of the Bay, where blooms of Enteromorpha on mud flats are now often reported.
Corophium do occasionally simply get sick and die, although not much is known about the diseases that afflict them. Undoubtedly, some of the short-term rapid declines in populations observed are attributable to outbreaks of bacterial or viral infection. More research has been carried out on their parasites, particularly those that are passed on to shorebirds. One such example is a trematode flatworm, Maritrema sp., a near kin to the parasite that causes Bilharsia or Schistosomiasis in humans. It also lives in a snail as its initial host. It then produces free swimming larval "cercaria" that leave the snail, seek out a Corophium and burrow into its body cavity, where they develop into yet another larval stage known as a "metacercaria". When an infected Corophium is eaten by a sandpiper, as large numbers of them eventually are, the metacercaria grow to adults in the bird’s intestine. The parasite’s eggs are eventually excreted along with the birds faeces. Foraging snails consume both faecal particles and eggs and thus the cycle starts another revolution. Under normal conditions, the number of parasites is usually very low and they do little if any harm to any of their hosts, a wise strategy on their part. However, under conditions of environmental stress, such as unusually high water temperature, the host’s resistance may be reduced, allowing parasite numbers to skyrocket. In Corophium this causes a form of anaemia that weakens and eventually kills them. Interestingly, it has been found that moderately infected animals crawl about more than uninfected ones, making them even more vulnerable to predation by shorebirds, which suits the parasite just fine. It is believed that many of the unexplained Corophium die-offs are probably a result of such outbreaks of parasites.
The quest continues Over the last two decades we have learned enough about Corophium volutator to affirm that it is indeed a "keystone species" on the mudflats of the upper Bay of Fundy. We have had some intriguing glimpses into many of the subtle ecological balances that exist between Corophium and its predators, its food and its glutinous habitat. It is this growing awareness of just how delicately balanced some of these relationships are that makes scientists increasingly fearful of the possible long-term consequences of human activities and developments to the Bay and its wildlife. There is still much to learn about Corophium and undoubtedly many important ecological linkages and balances that we are completely unaware of. To promote co-operation among the many researchers working on different parts of this complex ecological jig-saw puzzle, the Bay of Fundy Ecosystem Partnership (BoFEP) and the Fundy Marine Ecosystem Science Project (FMESP) have formed a Corophium Working Group comprising a dozen or so members. They meet periodically to review their results and discuss ideas for new research. They are also assembling a large database of information about the animal. They are hoping to construct different types of models that will mimic the ecological interactions between Corophium and its habitat, prey and predators. This will help them to predict how the populations might respond to changing environmental conditions, be they natural or a result of human activity. The models will also help to pinpoint what we still don’t understand about the biology and ecology of the elusive Corophium and thus guide the researchers’ future efforts. Their evident enthusiasm and dedication to the quest ensures that we will continue to see flocks of researchers foraging on the tidal flats of Fundy in search of more tiny pieces to the grand puzzle. What they learn will advance our efforts to ensure that Corophium populations continue to thrive and sustain the fish and birds that depend on them. So long as Corophium are abundant, those other foragers of the flats, the sandpipers, will keep returning in their millions in search of sustenance on their arduous southerly migrations, and will continue to dazzle and delight us with their spectacular precision aerobatics. Further ReadingBiology of Corophium volutator (Crustacea: Amphipoda) and the influence of shorebird predation on population structure in Chignecto Bay, Bay of Fundy, Canada. D.L. Peer, L.E. Linkletter and P.W. Hicklin, Netherlands Journal of Sea Research. Volume 20, number 4, pages 359-373. (1986).
The behaviour of Corophium volutator (Crustacea, Amphipoda). P.S. Meadows and A. Reid. Journal of Zoology, London. Volume 150, pages 387 to 399. (1966).
A selected bibliography on Corophium spp, with special emphasis on Corophium volutator and the wildlife ecology of mudflats in the Upper Bay of Fundy. Draft 2, Manuscript in preparation. P.G. Wells. Environmental Conservation Branch, Environment Canada, Dartmouth, Nova Scotia, 60 pages. (March, 1998).
Population dynamics and nutrition of Corophium volutator (Pallas) in the Cumberland Basin (Bay of Fundy). M.H. Murdoch, F. Barlocher and M.L. Laltoo. Journal of Experimental Marine Biology and Ecology. Volume 103, pages 235 to 250. (1986).
Male amphipods (Corophium volutator [Pallas]) show flexible behaviour in relation to risk of predation by sandpipers. J.S. Boates, M. Forbes, M. Zinck and N. McNeil. Ecoscience Volume 2, number 2, pages 123 to 128. (1995).
The reproductive biology of Corophium volutator and C. arenarium (Crustacea: Amphipoda). J.D. Fish and A. Mills. Journal of the Marine Biological Association of the United Kingdom, Volume 59, pages 355 to 368. (1979).
The Fundy Issues Series is an initiative of the Bay of Fundy Ecosystem Partnership. These publications describe our present scientific understanding of some of the environmental issues confronting the Bay. We hope that they will enhance your understanding of the biological richness and complexity of this unique marine area and the problems confronting it. Such awareness may encourage you to help in protecting it for the use and enjoyment of all, so that future generations may also share and appreciate its bounty and rare beauty.
This fact sheet may be reproduced and circulated, with credit to the
Bay of Fundy Ecosystem Partnership The Fundy Issues series is financially supported by:
The Environmental Conservation Branch
Environment Canada - Atlantic Region
Dartmouth, Nova Scotia
Written and produced by
J.A. Percy,
Sea Pen Communications
Granville Ferry. N.S.
Tel: (902)532-5129 e-mail: bofep@auracom.com