|
Fundy Issue #27 Spring 2006
Parlous POPs: Persistent Organic Pollutants in the Bay of Fundy
�The POP's of concern are
synthetic chemicals that never existed in the natural world; |
|
A
Cornucopia of Contaminants
Pernicious
POPs Researchers studying the structure and function of natural hydrocarbons soon learned that in the laboratory they could be manipulated and transformed into other chemical compounds with new and unusual properties. The POP's of concern are synthetic chemicals that never existed in the natural world; they were created by human ingenuity within the past century. They include some of our most economically valuable compounds but also some of the most dangerous to living organisms. For example, by replacing some of the hydrogen atoms in a natural hydrocarbon with chlorine atoms, chemists created an array of new and novel chemical compounds collectively called "chlorinated hydrocarbons". By the mid 1900s, this discovery had launched what some scientists call the "organochlorine era". Chlorinated hydrocarbons such DDT, chlordane, lindane, toxaphene and a host of similar pesticides promised to virtually eradicate unwanted insects and other pests threatening our health, crops and forests. Most POPs, including chlorinated hydrocarbons, possess another property that enhances both their usefulness and their danger. They are very persistent or long lasting in the environment, far beyond the time of activity needed to control pests. Most natural hydrocarbons degrade quickly to basic elements by normal bacterial activity and other natural processes. However, the presence of chlorine, bromine or similar elements in their structure makes POPs more stable and resistant to breakdown by normal biological processes. The new chemical pesticides are remarkably effective, and dangerous, because of their insidious nature, their ready absorption by organisms and their toxicity at low concentrations. The physiological processes of the pest organisms readily take up, process, transform, metabolize and excrete a wide array of natural hydrocarbons. The pesticide molecule, with its unfamiliar chlorine atoms, is just sufficiently similar to a natural hydrocarbon that it is taken up and treated as such by the organism's metabolic machinery. However, the intrusive chlorine disrupts the critical biochemical reactions and the physiological processes dependent on them, causing the organism to sicken and die. What the insect naively accepts as a typical, easily metabolized hydrocarbon is in reality a deadly Trojan horse. The organic nature of these pesticides, along with their "3P properties" (persistence, partitioning, potency), are what makes them so effective and so deadly. However, what was not fully recognized when these chemicals were indiscriminately and widely used in the 1940s, 50s and 60s was that their toxicity is seldom limited to insects or weeds. Most are so called "non -selective" toxicants and all living things, including humans, are vulnerable to some degree.
Partitioning
POPs POPs may enter the tissues in a variety of ways. They can be absorbed from the water through membranous tissues such as the skin, gills and lining of the gut, or be extracted from the air in the lungs. POPs in an animal's food may be readily absorbed into the body through the lining of the digestive system and either metabolized or stored in body fats. A large quantity of lipid is required for the production of eggs, the development of embryos, and in the case of mammals, the production of milk to suckle young. Hence, these are important ways in which POPs can be transferred from a female of some species to her young. This was clearly shown by studies of DDT in harbour porpoises in the Bay of Fundy. Concentration of DDT in the blubber of males increased with age, as they consumed more and more of it with their food. However, in female porpoises the amount decreased with age because they transferred much of the DDT to their fetuses and calves; pregnant and lactating females had reduced levels of DDT while calves had high levels.
POPs
in Populations It is particularly worrisome that many POPs are carcinogens and induce cancerous growth in various tissues and organs. Others are teratogens and cause the body's developmental processes to go awry, producing dysfunction and malformation of parts of the body during development and growth. Furthermore, critical biological processes in living organisms, such as growth, development and reproduction, are regulated by chemical messengers, called hormones, which are released into the blood stream at appropriate times by organs or tissues. These circulating messengers attach to specific receptor cells of tissues or organs elsewhere in the body, causing them to respond in a very specific way. These receptors respond only to molecules with a particular structure. Such integrated biochemical control mechanisms involving hormones are called endocrine systems. Many scientists are concerned by the discovery that some POPs being released into the environment are sufficiently similar in structure to some hormones that, once they get into the tissues of organisms, they fool the hormonal receptors into responding to them as if they were the corresponding hormones. They may bind to the receptor sites and cause a physiological response at an inappropriate time. Or, they may attach to the receptor and block the attachment of normal hormone molecules, although their slight structural differences prevent the receptor from launching the usual physiological response. Either way, the resulting effects on endocrine regulated biological processes such as growth, development or reproduction can be severe. Studies so far have only looked at a small number of possible endocrine-disrupting compounds acting on a few types of organisms; the overall ecosystem-wide threat is thus far from clear. Nevertheless, the detection of low levels of more and more potentially endocrine disrupting chemicals in our coastal waters is cause for great concern, if they are being bioaccumulated by organisms across the food webs.
Production
of POPs
Which human activities have introduced such significant quantities of so
many POPs into our global environment? The most obvious is the spraying,
from the air or from the ground, of large areas of our forests and
farmlands with various toxic mixtures to control a wide range of fungal,
plant and insect pests. Some of these chemicals eventually find their way,
directly or indirectly, into soil, lakes, rivers and coastal waters. Some
industries are also still releasing large amounts of POPs into the air or
water, even after spending millions on waste treatment. These include pulp
and paper mills, producers of chlorinated chemicals, manufacturers and
users of dry cleaning compounds, and producers of other products that use
halogenated hydrocarbons in their manufacturing processes. An example of
the latter is the widespread use of polybrominated diphenyl ethers (PBDEs)
as a flame retardant in products such as upholstered furniture. More
rigorous safety regulations enacted in recent decades have resulted in a
growing use of these compounds to reduce the flammability of many
household and industrial products. Recent estimates suggest that about
70,000 tonnes of PBDEs are produced in North America each year. The
quantities in the environment have risen steadily in recent decades and
have been detected in soil, water, air, fish, birds and marine mammals and
are also being absorbed by humans from their food. The production,
refining and use of crude oil and its various products also introduces a
variety of POPs, particularly PAHs into the environment. In addition, the
incineration of household, medical and industrial wastes releases a
variety of POPs.
A
Plethora of POPs In addition, most of the pesticides sold commercially and dispersed into the environment are not pure compounds. They contain varying amounts of so-called "inert ingredients" designed to make the product mix better, spray easier, last longer on the shelf, smell better, etc. Furthermore, even the principal "active ingredient" typically consists of a variable mixture of closely related isomers and congeners, largely because of the difficulty and expense of purifying large quantities of the material. For example, "aldrin" is the ISO common name for the commercial insecticide that has at least 95% of the pure active compound, which is HHDN (hexachloro-hexahydro-dimethanonaphthalene). In addition, chemical companies frequently use a registered trademark or "brand name" in marketing their own particular formulation of a commercial pesticide, whose specific composition may change from time to time and is proprietary information. Understandably, it is virtually impossible to fully assess the biological safety of such complex mixtures.
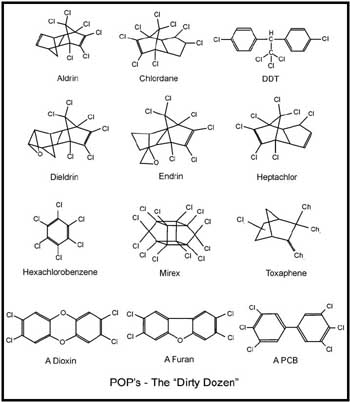
The
Dirty Dozen Far too many different POPs are being released into the environment to be able to conduct meaningful studies on the biological effects and regulate the inputs of most of them. The UNEP "International Negotiating Committee on POPs" has tried to narrow the field to a more manageable number by identifying a "dirty dozen" (see figure), widespread chemicals that may pose the gravest threats to ecosystems and human health. The list includes several common pesticides such as aldrin, chlordane, DDT, dieldrin, endrin, heptachlor, mirex and toxaphene; chemicals used industrially such as hexachlorabenzene, and PCBs; and unintended byproducts such as dioxins and furans. Unfortunately, this initial list is rapidly being overtaken by the constant discoveries of new persistent industrial compounds in the environment.
Fundy
Findings
Sediments
and shellfish
-
Measurements on samples of sediments collected at many different
places reveal that many POPs, including pesticides, PCBs and PAHs, are
present in the environment throughout the Gulf of Maine and Bay of Fundy.
Generally, the concentrations are higher nearer to the coast than further
offshore and tend to be significantly elevated near large cities and
industrialized areas and are therefore much higher in the southern Gulf
than in northern areas and the Bay of Fundy. A similar north-south
gradient is also evident in the results of the ongoing Gulfwatch Program
which periodically measures selected contaminants in a sentinel organism,
the blue mussel, collected from specific sites all around the Gulf of
Maine. Gulfwatch is described in more detail in Fundy Issue #12:
"Gulfwatch: Putting a Little Mussel in
Gulf of Maine Monitoring". The Gulfwatch results indicate that the
amounts of the principal types of POPs are mostly below Canadian and U.S.
guidelines intended to protect wildlife populations and human health.
Exceptions include areas near large industrial centres or major ports and
for a period in areas around a large oil spill. The Gulfwatch database
does not extend back to the 1950s and 60s, the time of peak production and
release of many POPs, nor does it yet cover a long enough period to
clearly demonstrate whether the concentrations of contaminants are now
declining. It does, however, provide a comprehensive quantitative snapshot
of present contaminant levels against which future changes can be
measured.
Seabirds - One of the first real warnings that chlorinated pesticides were having a devastating effect within ecosystems were reports in the 1950s and 60s, summarized in Rachel Carson's Silent Spring, that large numbers of birds were dying in pesticide treated areas. In addition, some species, particularly raptors at the top of the food chain such as hawks and eagles, weren't reproducing because the developing embryos were dying or the eggshells were so thin that they broke too easily. Female birds transfer a large portion of their lipids, along with any contaminants present in them, to their forming eggs. This makes bird eggs a standard, easily sampled indicator of relative concentrations of POPs and other pollutants in the surrounding environment. In 1968, Environment Canada began monitoring several contaminants, including POPs, in the eggs of a number of species of seabirds collected periodically from colonies around the Maritimes. The amounts of POPs found in different species usually varied with their diet and level in the food chain. For example, gannets and cormorants that eat fish (herring, mackerel etc.) had higher concentrations than terns that feed on zooplankton. An exception was an elevated level of several POPs in plankton-eating storm petrels. This may be because they often feed at the sea surface where lipophilic contaminants accumulate in the very thin, naturally oily surface film. This ongoing sampling and analysis of seabird eggs in eastern Canadian waters over five decades clearly showed the geographic distribution of contaminants, their high and often toxic concentrations in some species in the 60's and early 70s, and their gradual decline as some chemicals, such as DDT and PCBs, were banned and the production and use of others were better regulated. Typically, organochlorine levels were highest in eggs from colonies around the Gulf of St. Lawrence, downstream from the major industrial centres of the continent, intermediate in those from the Bay of Fundy region, and lowest in eggs from coastal Newfoundland and Labrador. The concentrations of many POPs, such as DDT, DDE and PCBs declined markedly in bird eggs in the Fundy region during the late 1970s and 80's, eventually reaching levels that stubbornly persist to this day, despite the bans and other restrictions on their use. This undoubtedly reflects an ongoing slow release of contaminants deposited in sediments as well as ones leaching from old landfill sites, in addition to the long-range atmospheric and biological transport of contaminants into the region. Other organochlorines, such as dieldrin and hexachlorobenzene, have not decreased significantly, while toxaphene has even increased. However, many scientists are confident that the amounts of POPs still present in seabirds and their eggs pose no immediate threat to their health and survival. Marine
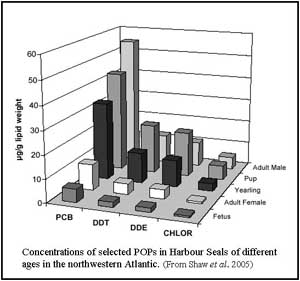
mammals - In the early 70's, the late David Gaskin and his students
from the University of Guelph collected blubber samples from harbour seals
from Boothbay Harbor, Grand Manan and Deer Island. They analyzed these for
chlorinated pesticides and PCBs. PCB levels were particularly high,
ranging from 32 to 240 parts per million. However, female seals had only
about a fifth of the POPs present in males, probably because of transfer
to the pups, which had elevated levels. More recent studies on seals in
the region reveal that while the levels of DDT (and its breakdown products
DDE and DDD) have declined significantly since the 1970s, PCB levels have
decreased only slightly. PCBs, DDT and chlordane presently account for 95%
of the POPs measured, while mirex, HCHs and dieldrin are present at much
lower levels and aldrin, endrins and methoxychlor are not detectable. The
concentrations of PCBs still present in the seals are sufficient to raise
concerns about possible effects on their reproduction or on their
endocrine and immune systems.
Gaskin and his students also looked at organochlorine pesticides and PCBs in 60 harbour porpoises collected in 1969-70 in the Fundy region. The DDT levels were much higher than in the seals. In fact, the concentration measured in males was the highest then reported for any wild marine mammal. Since DDT use in the region had been curtailed in 1967, the researchers decided to continue sampling for several more years to see if they could detect evidence of a decline in the contaminant present in the tissues. Between 1969 and 1973 the DDT levels in tissue went "down very remarkably". However, between 1974 and 1977 the average concentration rose almost to the earlier maximum. It may be that significant amounts of DDT were being resuspended from bottom sediments. DDT levels subsequently decreased again and there has been an overall decline from the 1970s to the 1990s. Early on, PCB levels in the porpoises were also high, particularly in the blubber. Between 1971 and 1977 there was no evidence of any decline in the concentration. By the mid 1990s, PCBs still formed the bulk of the POPs present in porpoise tissue. The persisting high levels in both seals and porpoises clearly warrants a closer look at possible effects on their health, especially as the PCBs are unlikely to decrease significantly for some time because of chronic inputs from industrial and municipal dumpsites. Many endangered North Atlantic Right Whales summer near the mouth of the Bay of Fundy. Part of the recovery plan for the species involves measuring contaminants in their tissues in order to evaluate possible health implications. The most comprehensive sampling program was carried out in the late 1980s. Small plugs of blubber were collected for analysis by means of a small, retrievable, harpoon-like, sampling tube fired from a modified crossbow. While many organochlorines were present in the blubber, their overall levels were
Curbing
the Contaminants Environment Canada must continue to gather data about the quantities of POPs and other chemicals being released into the environment by individual industrial plants and other economic operations such as forestry, mining and farming, and to make this information publicly accessible via the National Pollutant Release Inventory. This not only provides industry with a yardstick to measure, and an incentive to make, progress in reducing contaminants, but also spotlights significant problem areas still remaining. As contaminant removal technology improves, new manufacturing procedures are developed and lower toxicity materials become available, the regulations under the Canadian Environmental Protection Act, the Fisheries Act, and the Pest Control Products Act need to be regularly revised accordingly. This may involve periodically tightening the national standards for permitted release of toxic substances and strengthening the codes of practice for different industrial sectors. There should also be a continuation of the research to better understand the biological effects of POP's as well as a comprehensive program to determine the toxicity of newly developed POPs before they are approved for commercial use. More field studies are needed to determine whether the levels of POPs now present in the marine environment are affecting plant and animal populations. This is particularly true for endocrine disrupting compounds that may be insidiously affecting reproduction and development of marine organisms. We also need to continue monitoring trends in the concentrations of selected POPs in the marine environment and in key indicator species from all around the Gulf of Maine. This will indicate the effectiveness of regulatory programs designed to reduce inputs and also detect emerging threats. The Gulfwatch Program has already demonstrated the benefits of such a long-term, geographically broad approach to monitoring. POPs can travel great distances with atmospheric and oceanic circulation and biological transport, and degrade ecosystems far from their point of origin. Much of the POPs now present in the Fundy watershed comes from the distant industrial heartland of North America and even farther afield. Therefore, provincial and federal governments must continue to actively negotiate regional, national and international agreements to reduce the amounts of POPs released into the global environment. UNEP, the United Nations Environmental Programme, is already working towards developing an international consensus on regulating many toxic chemicals. The Stockholm Convention on Persistent Organic Pollutants POPs, adopted in 2001, "seeks the elimination or restriction of production and use of all intentionally produced POPs (i.e. industrial chemicals and pesticides) and, when feasible, ultimate elimination of releases of unintentionally produced POPs such as dioxins and furans". The Convention also requires that "stockpiles must be managed and disposed of in a safe and environmentally sound manner". The eventual goal is the elimination of the use of the pesticides aldrin, chlordane, dieldrin, endrin, heptachlor, hexachlobenzene, mirex and toxaphene. The use of PCBs by industry will also be discontinued. However, DDT may continue to be used in certain countries to kill disease-carrying insects until a safer, cost effective control method is found. The Stockholm Convention builds upon earlier treaties on the management of hazardous chemicals, particularly the 1989 Basel Convention on the Control of Transboundary Movements of Hazardous Wastes and their Disposal and the 1998 Rotterdam Convention on the Prior Informed Consent Procedure for Certain Hazardous Chemicals and Pesticides in International Trade. This suite of UNEP Conventions forms an international framework for "lifecycle" or "cradle to grave" management of the major types of hazardous compounds, particularly POPs, by implementing such management concepts of "Best Available Techniques" and "Best Environmental Practices". Many nations, including Canada, have already ratified these conventions. However, the difficulties and challenges associated with destroying the remaining stockpiles of various POPs, enforcing the more rigorous regulations, monitoring older POPs still in the environment, and the continuing production of new types of POPs means that toxic chemicals will be an ongoing environmental issue. In addition, new studies revealing more and more insidious sublethal effects of these compounds on marine organisms and humans adds even greater urgency to international efforts to control their release into the environment. However, we cannot rely solely on government agencies, international conventions or more stringent regulation of industrial inputs to eliminate the threat from POPs. Many industries have significantly reduced their emissions in recent decades and continue to seek less problematic chemicals for manufacturing processes. Ironically, the relative proportion contributed by non-commercial and domestic sources has been steadily rising because expanding urban populations, a broadening range of household and garden products containing hazardous compounds, coupled with inadequate regulation of their use and disposal, have made the general public collectively a significant polluter. We need to educate ourselves about the dangers of indiscriminately spreading toxic chemicals on our properties and thoughtlessly flushing leftover chemical wastes and potent cleaning agents down our drains. We need to be more aware of the need for proper and safe storage, handling, mixing, application and disposal of the toxic chemicals that are so readily available over the counter at local hardware stores and garden centres. We particularly need to read and heed the informative warning labels that government regulations require on all such hazardous products. Everyone who lives in the Bay of Fundy and Gulf of Maine watersheds has an important role to play in protecting our coastal ecosystems, and ourselves, from the worrisome scourge of toxic POPs. |
Further Information Silent Spring. Rachel Carson. Houghton Mifflin, Riverside Press, Cambridge, Mass. 368 pages. (1962). Contaminants in Canadian Seabirds. State of the Environment (SOE) fact sheet number 90-1. Environment Canada - Conservation and Protection, Ottawa, ON. 12 pages. (1990) Mercury, DDT and PCB in harbour seals (Phoca vitulina) from the Bay of Fundy and Gulf of Maine. D.E. Gaskin, R. Frank, M. Holdrinet, K. Ishida, C.J. Walton and M. Smith. Journal of the Fisheries Research Board of Canada 30(1), 471-475. (1973). Chemical Contaminants in Canadian Aquatic Ecosystems. R.C. Pierce, D.M. Whittle and J.B. Bramwell (eds.). Department of Fisheries and Oceans Canada. Canadian Government Publishing, Ottawa ON. 329 pages. (1998). Polychlorinated biphenyls and chlorinated pesticides in harbor seals (Phoca vitulina concolor) from the northwestern Atlantic coast. S. D. Shaw, D. Brenner, A. Bourakovsky, C.A. Mahaffey and C. R. Perkins. Marine Pollution Bulletin, volume 50, number 10, pages 1069 to 1084. (2005) Health of the Oceans: A Status Report on Canadian Marine Environmental Quality. P.G. Wells and S.J. Rolston (eds.). Environment Canada, Ottawa ON and Dartmouth NS. 187 pages. (1991). The Chemical Environment of the Bay of Fundy. P.G. Wells, P.D. Keizer, J.L. Martin, P.A. Yeats, K.M. Ellis and D.W. Johnston. Pages 37 to 61 in Bay of Fundy Issues: A Scientific Overview. (J.A. Percy, P.G. Wells and A.J. Evans (eds.) Environment Canada - Atlantic Region Occasional Report number 8. (1997). Websites Greenfacts.org page on dioxins: http://www.greenfacts.org/dioxins/l-2/dioxins-1.htm Environment Canada National Pollutant Release Inventory: http://www.ec.gc.ca/npri-inrp-comm/ International Standards Organization (ISO) Compendium of Pesticide Common Names: http://www.hclrss.demon.co.uk/ Stockholm Convention on Persistent Organic Pollutants (POPs): www.pops.int United Nations Environmental Programme site on POPs: http://www.chem.unep.ch/pops/default.html US EPA Pesticides information site: http://www.epa.gov/pesticides/a-z/index.htm
This Fundy Issue is
financially supported by: The views expressed herein are not necessarily those of Environment Canada or other BoFEP partners.
Written
and designed by J.A. Percy,
Acknowledgement |
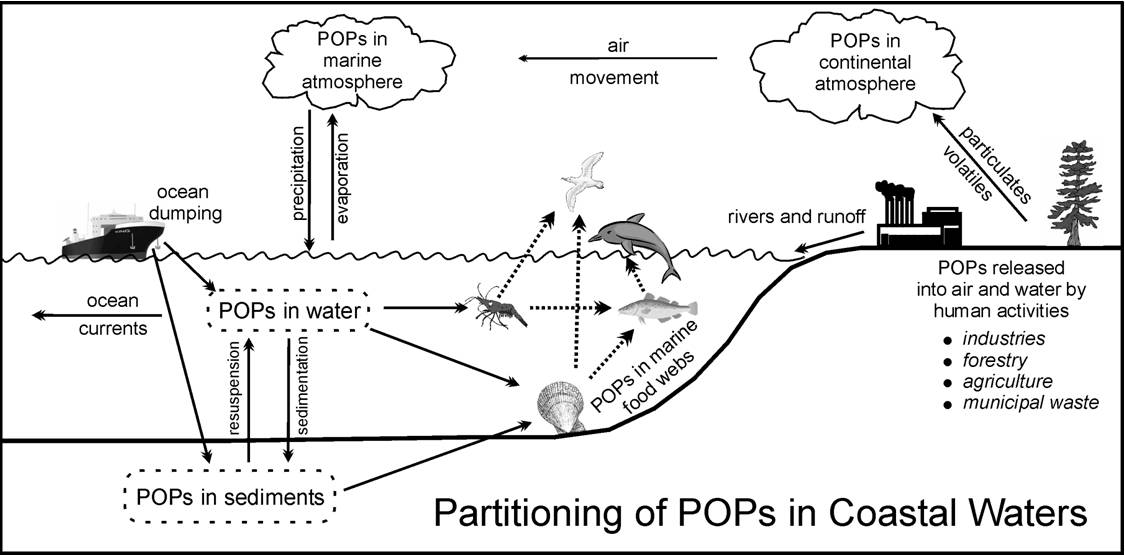
 POPs are readily
taken up by marine organisms and incorporated into the ocean's food
chains. This is of great concern, not only because the chemicals can harm
marine plants and animals and upset the productivity and integrity of
ocean ecosystems, but also because of the potential threat to human health
from contaminated seafood. Most POPs interfere with many metabolic
processes of cells and tissues, causing an exceptionally wide range of
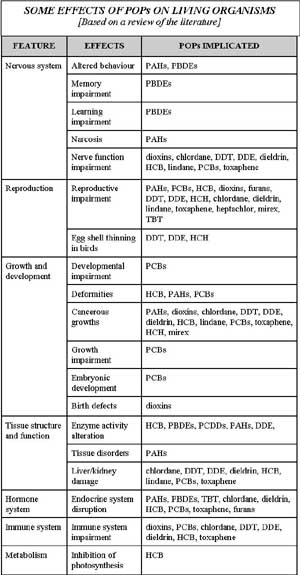
biochemical, physiological, behavioural and clinical problems (see table).
Factors that influence their toxicity to organisms include their
concentration in the environment, the ease with which they are taken up by
different tissues, the length of exposure, the manner in which they
interact with biochemical and physiological processes, and the ability of
organisms to break down and excrete them.
POPs are readily
taken up by marine organisms and incorporated into the ocean's food
chains. This is of great concern, not only because the chemicals can harm
marine plants and animals and upset the productivity and integrity of
ocean ecosystems, but also because of the potential threat to human health
from contaminated seafood. Most POPs interfere with many metabolic
processes of cells and tissues, causing an exceptionally wide range of
biochemical, physiological, behavioural and clinical problems (see table).
Factors that influence their toxicity to organisms include their
concentration in the environment, the ease with which they are taken up by
different tissues, the length of exposure, the manner in which they
interact with biochemical and physiological processes, and the ability of
organisms to break down and excrete them.  POPs enter the
environment from many different sources. Prior to the industrial
revolution there was a steady input of a relatively small number of
different POPs, such as the PAH's (polyaromatic hydrocarbons) that are
produced by natural processes, such as volcanic activity, crude oil seeps
and forest fires. While such natural inputs continue, they are now
generally small, both in the quantities and the variety of different
chemicals, compared to the contributions from human activities. The
concentrations of POPs in the environment are typically very much higher
near large industrialized urban centres compared to remoter rural areas.
For example, in the Gulf of Maine, the Gulf Watch mussel-monitoring
program reveals that the levels of some POPs (as well as other toxic
chemicals) are much higher in coastal waters of the southern Gulf than in
the northern Gulf and Bay of Fundy. However, currents of air and water can
carry significant quantities of many POPs over most of the globe, even to
the remotest places with no local human inputs.
POPs enter the
environment from many different sources. Prior to the industrial
revolution there was a steady input of a relatively small number of
different POPs, such as the PAH's (polyaromatic hydrocarbons) that are
produced by natural processes, such as volcanic activity, crude oil seeps
and forest fires. While such natural inputs continue, they are now
generally small, both in the quantities and the variety of different
chemicals, compared to the contributions from human activities. The
concentrations of POPs in the environment are typically very much higher
near large industrialized urban centres compared to remoter rural areas.
For example, in the Gulf of Maine, the Gulf Watch mussel-monitoring
program reveals that the levels of some POPs (as well as other toxic
chemicals) are much higher in coastal waters of the southern Gulf than in
the northern Gulf and Bay of Fundy. However, currents of air and water can
carry significant quantities of many POPs over most of the globe, even to
the remotest places with no local human inputs.